An isotopic topic
The 118 elements in the periodic table are uniquely defined by their atomic numbers, and this invariantly refers to the number of protons in the nucleus of an atom of that element. Hydrogen 1, Helium 2, Lithium 3...bla bla... Platinum 78, Gold 79, Mercury 80 ... bla bla .... Ununoctium 118, and there may well be a 119th and 120th coming along shortly, although only the first 94 elements are ever found naturally. The high-faluting ones are synthesized at vast expense and typically last for milliseconds. Only three atoms of Ununoctium have ever been synthesized. If you ask "Why would they do that?" their answer would be "Because now we know." By the way, Ununoctium officially became an element only four months ago. How time doesn't fly.
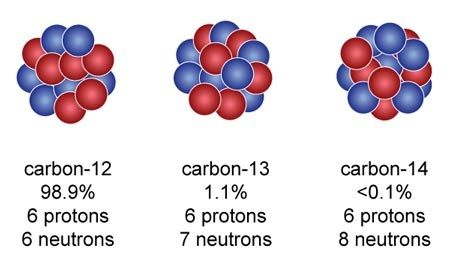
Counting protons does not tell us all about the nuclear structure of an element, because in almost every case the nucleus is made of neutrons as well as protons. Take carbon (please...) Its atomic number is 6, so its nucleus always has 6 protons by definition. But 98.9% of the carbon on Earth also has 6 neutrons in its nucleus. 6 + 6 = 12, so 12 is what is now called the mass number of normal, bog standard carbon. Chemists know it by its nickname, 12C6. But about 1% of carbon atoms you might find lying around have one extra neutron, and a very small proportion have two extra neutrons. They are, of course, 13C6 and 14C6 respectively, and the three different carbons are the isotopes of carbon.
credit: emaze
Now we get to the radioactive part. Put a teaspoonful of carbon-12 or carbon-13 on your kitchen table, and they'll stay right there for ever. They are stable isotopes. Even if you walk away and come back in a million years, there will still be exactly the same number of atoms on the table (unless you've had a fall of soot.) Carbon-14, however, is not like that. It is unstable, and spontaneously decays. Radioactive decay takes several forms but in this case each decay event causes a neutron to turn into a proton. The mass number thus remains 14, but the atomic number goes up one, and therefore that atom is no longer carbon. It becomes the next element in the table, which happens to be nitrogen. This decay process has a half-life of 5,730 years, so after that length of time half your carbon-14 has turned into nitrogen-14 and literally vanished into thin air. Nitrogen itself has one other stable isotope, nitrogen-15, plus a raft of unstable ones. Some of these decay back to carbon, some go on up to the next element, which happens to be oxygen.
So there's yer basic tute on what isotopes are and what mischief they get up to. Enter xenon.
Noble xenon
Helium, neon, argon, krypton, xenon, and radon are the six so-called "noble" gases, although -- isn't this interesting? -- the brand new element Ununoctium may well turn out to be an ultra-heavy noble gas. The big feature of the nobles is that they don't form bonds with other elements, unlike carbon which will make molecules with anything that asks, the slut. So there's no such thing as argon hydroxide or xenon hydrochloride, for example. Xenon has the distinction of having more stable isotopes than any other element except tin. Here's a reference to 13 xenon isotopes, eight of which are stable. "NA" means "Natural Abundance."
====================================
124Xe - NA 0.095%, decays to 124Te, half-life 5 x 1016 y (essentially stable)
125Xe - NA zero, decays to 125I, half-life 16.9h
126Xe - NA 0.089%, stable
127Xe - NA zero, decays to 127I, half-life 36.345d
128Xe - NA 1.91%, stable
129Xe - NA 26.4%, stable. Produced by beta decay of 129I, half-life 16 million y
130Xe - NA 4.07%, stable
131Xe - NA 21.2%, stable. Fission product of Thorium, Uranium, and Plutonium. Can be a decay product of 131I or 131Cs
132Xe - NA 26.9%, stable. The most common natural isotope. Can be a decay product of 132I or 132Ba
133Xe - NA zero, decays to 133Cs, half-life 5.25d
134Xe - NA 10.4%, decays to 134Ba, half-life 1.1 x 1016 y (essentially stable). Fission product of Thorium, Uranium, and Plutonium
135Xe - NA zero, decays to 135Cs, half-life 9.14h. Fission product of Thorium, Uranium, and Plutonium
136Xe - NA 8.86%, decays to 136Ba, half-life 2.2 x 1021 y (essentially stable). Fission product of Thorium, Uranium, and Plutonium
====================================
The four isotopes that are fission products can be expected to be unusually plentiful in the aftermath of a nuclear fission weapon detonation, and indeed these isotopes have been used to detect weapon tests. Xenon-129 is not a fission product in the conventional sense, but it is generated by a special nuclear reaction called fast fission. Fast fission is used in advanced reactor designs and in some fusion weapons (H-bombs, to put it crudely.)
Interplanetary warfare?
As shown in the above reference, in planet Earth's atmosphere, xenon 129 is only slightly less abundant than the dominant isotope 132 -- 26.4% cf. 26.9%. Thanks to some brilliant space science, we now know a good deal about the atmosphere of planet Mars, and in that atmosphere 129 is more than 2.5 times as abundant as 132 (although both, of course, are far less abundant in absolute terms than the terrestrials.) This has led a maverick physicist called John Brandenburg to speculate that at least one fast fission event -- such as the detonation of a thermo-nuclear weapon -- has occurred at some time in Mars' history. Brandenburg has attracted quite a bit of attention by publishing this conjecture -- he's become a favorite on the pseudoscience circuit that includes late-night radio chat shows, internet radio, and TV productions with a very lenient view of what constitutes a historical fact (Yes, Ancient Aliens, I'm talking about you.)
Figure from John Brandenburg's 2015 LPSC poster. NOTE that these data are relative, not absolute
So is Brandenburg right? Was Mars nearly obliterated by nuclear warfare? I hesitate to go up against a well-qualified physicist but my answer is probably not. Very probably not. The thing is, you don't need such a radical hypothesis as fast fission to explain the 129/132 isotope ratio on Mars. Look at that reference table again. Xenon-129 is remarkable among the isotopes in that it can be, and is, created by decay of Iodine-129 with a half life of 16 million years. Is Iodine-129 itself a fission product? No it is not. It is, however, a solid, not a gas, at normal temperatures (boiling point 184.3 °C.) So if, let's say, Mars' primordial atmosphere was almost entirely lost due to some catastrophe early in its history, iodine would not be affected and it would not be strange that xenon-129 would be preferentially replenished.
Well, that's exactly what Marsologists say happened, some time within the first 100 million years after the planet was formed. The planet has been around for some 4.5 billion years, so there has been plenty of time for iodine-129 to quietly turn itself into xenon-129, even given that 16m year half-life. Iodine-132 also decays to xenon-132, but that isotope is extremely rare and the decay half-life is measured in hours, so it's irrelevant.
Wrap-up
NASA gets way too much scorn these days from people who are too ignorant to know better than to believe the chief NASA-haters, Richard Hoagland and Mike Bara. The agency is accused of fudging and masking important data, and avoiding key questions about extraterrestrial life. The truth, from what I know and have observed, is that NASA might delay release of some planetary data in order to give priority to the science teams to peer-review and publish them, but there is absolutely no concerted policy to hide evidence of living things or dodgy guesswork like nuclear explosions on Mars.
There is too much scorn, and not nearly enough gratitude for the brilliance of the JPL designers whose experiments make it possible to have these arguments.
With grateful thanks to my excellent research assistant, Google Search.
Update 8 May 2016
Element 118 has now been provisionally named Oganesson (Og), honoring Yuri Oganessian who led the team that synthesised element-117.
=====================================
References:
Evidence for large, anomalous, nuclear explosions on Mars in the past. --Brandenburg, J.E. 46th Lunar and Planetary Science Conference (2105) poster session
Rational wikipedia article on Dr John Brandenburg
Was Mars murdered? Podcast shownotes by Dr Stuart Robbins,
Wikipedia article on xenon and its isotopes
Wikipedia article on Martian atmosphere
Fission product tables for typical heavy isotopes. IAEA document
USGS Isotope resource - Xenon

48 comments:
Theadora wanted to draw our attention to something Richard Hoagland is doing. I'm not willing to have the URL posted here. Hoagland is very much part of the problem.
The following pareidolic image is often used by the nuclear explosion crowd as proof of such. It did the rounds last year.
http://i1.mirror.co.uk/incoming/article5318427.ece/ALTERNATES/s615b/cloud-1.jpg
However, Metabunk and others have shown how it IS just pareidolia:
https://www.metabunk.org/debunked-nuclear-cloud-on-indian-mars-orbiter-image.t5876/
I was just letting you know, Patrick, where you can listen to him for free at his new network, so you can resume your reviews of his shows, ass you used to doo before you had to pay for the privilege.
That was not news to me. The problem is that I'm always in bed at that time.
Well, then I guess you still, won't have Dick to kick around anymore?
@expat
Ripley doesn't like you very much now does she :-)
....and she doesn't answer tough questions either :-)
what does that say......?
Adrian
Theadora: You obviously missed this, then, which I posted just 10 days ago.
Adrian: I'm gutted, man. Sharper than a serpent's tooth it is to be rejected by a blow-up doll.
What tough questions don't I answer? Nobody ever asks me anything. If I missed a question directed to me, please, direct me to it and I'll cram the answer back up your ass. I've got nothing against Patrick. Whenever he censors me, he always lets everybody know, so that they can go to my page and see what it was.
Clicked on the TEM main website and it's not there right now, Bluehost comes up. Are they moving or is it gone? A nice make-over with at least HTML 4.01 would be great ;) Was it mentioned anywhere? Too lazy to check right now. Stay tuned...
Dee
I noticed that yesterday Dee. Inspection of whois records shows that the domain registration is paid up til December 2017 (In Robin Falkov's name) so this can only mean that they haven't paid Bluehost's invoices.
Last July Hoagland promised that the site would be fully updated. So much for that.
Just because nobody ever asks me anything, though, doesn't let anyone off the hook. I'll still let you know what's what.
othersideofmidnight, now forwards to: theothersideofmidnight.
Rowland must be holding Hoagland's websites, hostage.
Domain Name: OTHERSIDEOFMIDNIGHT____
Registrant Name: KEITH ROWLAND
Registrant Organization: ROWLAND NETWORK COMMUNICATIONS, LLC
Registrant City: MESA
Registrant State/Province: AZ
Registrant Postal Code: 85205
Registrant Country: US
Registrant Phone: +1.4805599937
Domain Name: THEOTHERSIDEOFMIDNIGHT____
Registrant Name: RICHARD HOAGLAND
Registrant Organization: ENTERPRISE MISSION
Registrant City: PLACITAS
Registrant State/Province: NEW MEXICO
Registrant Postal Code: 87043
Registrant Country: UNITED STATES
Registrant Phone: +1.5058676237
Any action on his fb page?
Chris, the content on his page is almost completely mirrored on the Other Side of Midnight page.
So it's an advertising page. Looked at the station line up and there seems to be a lot of other hosts of questionable value. Is the station part of some network like Premier?
Chris: The main point is that KCAA rents its chat-show facilities to anyone who can pay.
The difference is that theothersideofmignight is written with a WYSIWYG (What You See Is What You Get) page editor, as easy to use as Blogspot, so that, that Dick Hoagland can do it himself instead of bargaining with Rowland.
It's easier than you think to obtain distribution of a radio or even television program. It's not like trying to land a job with the BB FLICKING C. Just pay for all the ad time, with the option of reselling. Find a low powered station, where the weak signal can manage to reach an adjacent large market. It will be cheap to pay for, but with potential to be heard by a big audience. Of course, if the station has Internet streaming, better yet.
radio-locator.com
So Hoagland is paying, not being paid. Why would he go for that?
The business model hasn't changed. He recruits members who pay to get access to the archives, and he can sell ad minutes. How successful that will be remains to be seen.
My guess is it will work as well as the whole selling of DVD's thing worked.
@Ripley - for example in reaction to your "...nobody ever asks me anything..."
you stated in the previous item "If anything from the future were to manage somehow to enter the past, the past would change, and annihilation would result. Therefore, there are no time travelers to the past and probably technology of time travel into the past will never be invented, because we are all still here."
The question was “your statement is based on the premise that "our concept" of time is a linear phenomenon.....It goes entirely above and beyond the fact that "we" invented" such a concept known as time and that somehow it has to be linear. If your statement turns out to be correct....you have to proof this "figment of [y]our imagination"
the floor is yours....:-)
Adrian
Well, Adrian, the Buddhist model that "all is illusion" is conveniently by taking a cast iron skillet and smacking into the side of your own thick skull. While it's true that I struggle to recall the previous day's lunch, I distinctly remember that there was a yesterday. The mere act of observing a Quantum partial is to change it. If you fuck with yesterday, then there is no today. Since there is a today, nobody changed the past. I find this more 'believable' than Buddhism. To prove it is a risk not worth taking. Of course the bozos who first split the atom reckoned that there was a pretty good chance that it would create a chain reaction that might destroy the entire Universe. Naturally, they lived for this shit, so said, "Eh, what the heck." Same with the Hadron Collider, which may create a black hole in place of planet Earth.
Although, more specifically to address the perception of time as being linear, in the Prophetic, Book of Revelation, the future is written of in past tense, while the past is written of in future tense. Dr Hal Lindsey has noted that it seems as if there are two streams of time which flow in opposite directions.
An error-filled commentary, as we've come to expect.
- It was the first H-bomb, not the first A-bomb, that gave rise to fears of non-containment.
- The fear was that it might destroy the planet, not the universe.
- There is not the slightest chance that the LHC will ever create a destructive black hole.
Okay, so the War Department was in too big a rush to consider the downside, but if the H bomb could split all the atoms in the World, then what would be to stop it from igniting the hydrogen of Outer Space? The Martians might get real, ticked off by something like that. Furthermore, I don't find your assurance that Earth won't become Swiss Cheese, less relaxing than a couple double shots of Bushmill's Single Malt.
<< Okay, so the War Department was in too big a rush to consider the downside...>>
No. Uranium 235, unlike hydrogen, is not ubiquitous. Sure, fast neutrons spill out of nukes but there's nothing out there for them to split. So no worries.
<< but if the H bomb could split all the atoms in the World, then what would be to stop it from igniting the hydrogen of Outer Space?>>
It couldn't split all the atoms in the world. It couldn't split ANY atoms, dearest Theadora. It's a fusion weapon, not a fission weapon. Also there's no hydrogen in outer space. There's no anything in outer space. Please stop polluting this blog with inaccuracies.
(from Wikipedia)
Outer space
is the void that exists between celestial bodies, including the Earth. It is not completely empty, but consists of a hard vacuum containing a low density of particles, predominantly a plasma of hydrogen and helium
Atmosphere of Earth
By volume, dry air contains 78.09% nitrogen, 20.95% oxygen, 0.93% argon, 0.039% carbon dioxide, and small amounts of other gases. Air also contains a variable amount of water vapor, on average around 1% at sea level, and 0.4% over the entire atmosphere
@ Ripley
all I read from you is a lot of ranting and abusive language....I still waiting for a more scientific approach
Adrian
« a low density of particles... »
Ultra-low density. Roughly one atom per cc. No chance of sustaining a fusion reaction at all.
Isn't anybody going to comment on xenon isotopes? Seems like nobody read it *sob*.
Dearest Adrian, your question isn't scientific; it's philosophical. I'm just applying common subjective methodology of debunking, such as that used by the likes of CSICOP agents. You know, the 'if there is a more simple, seemingly plausible explanation then no proof is necessary,' sort of thing.
@Ripley
Interesting concept....So you can say and/or rant in all directions but there doesn't have to be a scientific validation and/or proof according to your way of perception and/or reasoning.
So to keep thing more simple....you stated "If anything from the future were to manage somehow to enter the past, the past would change, and annihilation would result."
Be a nice girl and proof it and stop horsing around darling.
Adrian
Expat, since you sobbed.
What about Brandenburg's linking of "his" Xenon to the presence of some stable isotope of Krypton (80) and "excesses" of uranium and thorium on the Martian surface? It would seem to me his theory is not just build around Xenon (but perhaps Helium hahah). To be honest I haven't looked very far into the subject but it seemed you were arguing for Brandenburg to be likely wrong by focusing on one isolated element of his theory. Wouldn't his line of reasoning be build around the simultaneous presence of multiple factors?
Dee: You're going to make me wish I hadn't sobbed.
I don't buy the "high concentrations of Th" idea. Yes, concentrations vary across the surface of the planet, but so do concentrations of all metals as proto-planets differentiate. By "high" we're talking ~1 ppm, as this map shows.
In his podcast, Stuart Robbins noted "a very, very length paper with over a dozen authors was published in 2007 using this as evidence for water carrying rocks to the lowest portion of the planet, draining into a northern hemisphere ocean, which is where the largest concentrations of potassium and thorium are."
I don't know about Kr-80. Thanks for the comment.
It's like Communism, Adrian. It can't be proven in a vacuum. The entire World must become a totalitarian state in order to see if it works or not. While I am being somewhat facetious, as my fundamental criticism of Patrick, and his buddies, is that they merely contraDICKt Hoagland, rather than prove him wrong. Of course, the way science works, is that one must at least qualify an untestable notion in a hypothetical manner, if not develop a testable theory, or have proven an experiment which has been duplicated by more than one peer. In all fairness, Hoagland should consider that his beliefs and assertions might not necessarily turn out to be accurate, and Patrick ought to at least be open to more potential possibilities.
As far as time travel goes, I will ad that I suspect that there is a natural barrier which prohibits a material return to the past, and that any attempt to send matter to a prior date, wouldn't annihilate existence, rather nothing would happen at all. It simply wouldn't work. That's an experiment which can be easily proven by any experiment you can manage to conjure up.
<>
Dear Theadora: Perhaps you should re-read this and this, for example.
Fair enough, Patrick, those are two examples of good work, but correct me if I'm also wrong that you likely accede to the predominate hypotheses that humans are descended from primates? If so, then is there any significant difference between that, and Hoagland's belief that man is descended from Spacemonkeys which evolved in caves on Mars?
Yes. The difference is that there is evidence for one and not the other. I assume you're just being playful here.
@Ripley
Darling....you're almost halfway answering answering a question in order to proof your statement "If anything from the future were to manage somehow to enter the past, the past would change, and annihilation would result."
with your latest remark "That's an experiment which can be easily proven by any experiment you can manage to conjure up."
So go ahead and proof it...what is keeping you...you still didn't proof your statement. Conjure up theory - test it rigorously and present us the law of Ripley. For the time being it is just an idea based on your opinion...and you know what they say about opinions according to Eastwood.
Adrian
Adrian, the experiment for proving my stated hypotheses, is already implicit in my previous comments. The theory would be that there already are a shitload of scientists attempting to go back in time, and that all of them will fail, or already have failed, without destroying all of existence. I just don't have a clue as to how we can get them to disgrace themselves by publicly admitting that.
Otherwise, I already have more than enough exercises in futility of my own, to keep me busy. Like trying to convince Gary Leggiere, that nobody in radio dares to accept Payola, nowadays.
That's pretty funny, Thea. But you know, Gene Steinberg might just go for it, Paracast isn't real radio.
Not to put too, fine a point on it, Patrick.
@Ripley
I see you are still horsing around but not a shred of legitimate proof for your stated hypotheses. Funny you mentioned the scientific term hypothesis....You know what it implies do you not?? A starting point for a theory which, if it turns out to be valid, will eventually lead to an explanation and/or a law. The only thing you have thus far are assumptions and you are nowhere near a possible theory...annoying but there it is.
The proof is in the pudding :-)
but in order to spice it up just little bit... Why not have a thought experiment.
Let's say you conjured up a gismo that when powered up is able to transport [or any other definition you might fancy] into a specific time in the past. Now...we have a number of baskets made of natural materials filled with apples [and not Jobs apples :-]. We send one to every century from the first century up to the twentieth century at random locations on land. How and why do these baskets full of apples annihilate everything and which basket full of apples does the first annihilation? And how does this relate to your concept of time....is everything annihilated all at once or will it be progressive and what would be the end result in terms of the law we know as conservation of energy?
Well then...snap to it
I said hypothesis specifically because I don't intend to develop a theory for me to test. I can't be trotting around all over, stamping out every little brush fire.
There use to be a belief system called, Darwinism. The term evolved into the so-called, Theory of Evolution, yet there was no testable theory proposed for it. Nowadays, the term is simply called, Evolution, as if it were a foregone conclusion.
Patrick accedes to Science as Democracy, whereas a preponderance of supposed scientists believe in hypothetical notions, accepting them as if having been proven.
The Scientific Method may be burdensome, but you aren't going to invent a time machine without it. I'm not trying to invent a time machine, and there is no incentive for me to prove that one can't be invented. I've stated my opinion, qualified it as such, and maintain my view. Of course, if anybody is foolish enough to fund the research, I'm willing to reconsider.
If so much as a single grain of sand from the present, were to somehow manage to be transported back into time, any point in time, there would be instantaneous annihilation of all existence. It would be quite, painless, no doubt. Perhaps if even so much as consciousness were to perceive the past, in real time, the same result would occur. There may be a natural barrier which prevents anything from the present or the future, of entering the past. Since we are all still here, time travel into the past never has happened, and it never will. I hope that expands and clarifies my hypothesis, for those who have been wondering.
There are examples of men who have transported out of body or perhaps just with mind, into the future, who have returned back to their original time in the past of that future which they visited, but Patrick has a history of censoring that information.
@Ripley
"If so much as a single grain of sand from the present, were to somehow manage to be transported back into time, any point in time, there would be instantaneous annihilation of all existence. It would be quite, painless, no doubt. Perhaps if even so much as consciousness were to perceive the past, in real time, the same result would occur."
the scientific observation and/or conclusion remains the same darling based on your [very] definitive i.e. "this is the bare truth" statements. Still no proof dear :-) to back up such bold and definitive statements and/or claims. Surely you realize that by now...
If this hypothesis of yours would hold a grain of truth then scientific method dictates and requires that one can test it and since your hypothesis produces a paradox which makes it "miraculously un-testable" you [un]consciously use this as an excuse to validate your point. It is a bit like stating that all Swans are white and if a black Swan would somehow appear in the habitat all Swans disappear instantly and It would be quite, painless, no doubt :-) so therefore all Swans are white.
Adrian
Even you seem to have an opinion, Adrian. That's an anecdotal observation, however, you actually might not even believe what you say, for all I really, know.
I could not find any references to significant amounts of Iodine 129 on Mars in any of the literature. Maybe I didn't look hard enough. Seems like Brandenburg is on to something here. It would be good to pursue this and look for more data on other isotopes to support or disprove the nuclear explosion hypothesis. It appears to me that if there was Iodine 129 decaying to Xenon 129, the Iodine 129 could have been produced by nuclear reactor operations on mars.
From Wikipedia - Iodine 129 is primarily formed from the fission of uranium and plutonium in nuclear reactors. Significant amounts were released into the atmosphere as a result of nuclear weapons testing in the 1950s and 1960s.
It is also naturally produced in small quantities, due to the spontaneous fission of natural uranium, by cosmic ray spallation of trace levels of xenon in the atmosphere, and some by cosmic ray muons striking tellurium-130
Thanks for the comment. You're correct--129I is not abundant in nature.
Post a Comment